Source: international ophthalmology news
Myopia is a worldwide public health threat. In the past few decades, the myopia rate in most parts of the world has been rising. Asia, especially East Asia, including China, Japan and South Korea, is the hardest hit area of myopia. Low concentration atropine eye drops is a new and effective clinical treatment for myopia, but the optimal concentration and long-term treatment of atropine have not been determined. Two previous studies of atropine in the treatment of myopia (atom1 and atom2) observed a "rebound phenomenon": after continuous treatment with 1%, 0.5% and 0.1% atropine for 2 years, myopia progressed faster after stopping treatment, but there was no rebound after treatment with 0.01% atropine. The results of atom2 study showed that based on the best balance between efficacy and safety, 0.01% atropine was the best concentration. But two years after atropine administration, should the treatment be continued or stopped? What is the rebound effect, long-term efficacy and optimal concentration after using 0.05%, 0.025% and 0.01% atropine? What factors are related to the rebound effect after low concentration atropine treatment? Is it safe to use low concentration atropine for a long time? To this end, Jason C. yam from the Chinese University of Hong Kong conducted a three-year study on the treatment of myopia progression (lamp) with three low concentrations of atropine. The aim of this study was to assess whether continued treatment (0.05%, 0.025%, 0.01% atropine) was better than stopping treatment in the third year; The long-term efficacy of continued treatment with these low concentrations of atropine for 3 years; Rebound effect after stopping treatment and its relationship with low concentration atropine. In the November 2021 issue of Ophthalmology magazine, the results of the third issue of lamp are fresh. Let's have a quick look.
research method
The lamp study included 4 to 12-year-old children with binocular myopia ≥ 1.0d and astigmatism < 2.5D, and the increase of myopia ≥ 0.5d in the past year in a double-blind, single center clinical trial.
In the first phase, children were randomly assigned to four treatment groups (0.05%, 0.025%, 0.01% atropine and placebo) and followed up every 4 months after initial treatment.
In the second phase, all children in the placebo group in the first phase switched to 0.05% atropine until the end of the phase. In this second year, children in the original atropine treatment group still received the same concentration of treatment.
In the phase III study of lamp, 350 children in the three original treatment groups (0.05%, 0.025% and 0.01% atropine) in the first phase were randomly divided into continued treatment subgroup and stop treatment or "exit" subgroup in a ratio of 1:1, and further stratified by gender and age (6-8 years old, 9-11 years old and 12-14 years old). For the subgroup that continued treatment, the subjects continued to receive the same concentration of eye drops into their eyes every night for the whole third year. The diopter and ocular axis (AL) of cyclic spherical equivalent (SE) were measured every 4 months.
1. The results of the third year of lamp showed that there were significant differences in myopia progression and ocular axis between the subgroups of continuing treatment and stopping treatment; Compared with 0.025% (0.22d) and 0.01% (0.18d) groups, the difference in myopia progression between the two groups was the most prominent in 0.05% (0.4d).
The mean myopia progression of children in the continued treatment subgroup (0.05%, 0.025% and 0.01% atropine treatment) was -0.28 ± 0.42d, -0.35 ± 0.37d and -0.38 ± 0.49d, respectively, while the respective al growth was 0.17 ± 0.14mm, 0.20 ± 0.15mm and 0.24 ± 0.18mm, respectively (Table 2). There was no significant difference in the progression of myopia in different concentration groups, but there was a large axial extension in higher concentration, with the axial extension of 0.33 ± 0.17mm, 0.29 ± 0.14mm and 0.29 ± 0.15mm respectively (P = 0.003).
The children in the stop treatment group were divided into 0.05%, 0.025% and 0.01% atropine treatment subgroups according to the atropine concentration used in the previous two years. The average myopia progression was -0.68 ± 0.49d, -0.57 ± 0.38d and -0.56 ± 0.40d respectively, and the corresponding axial growth was 0.33 ± 0.17mm, 0.29 ± 0.14mm and 0.29 ± 0.15mm respectively.
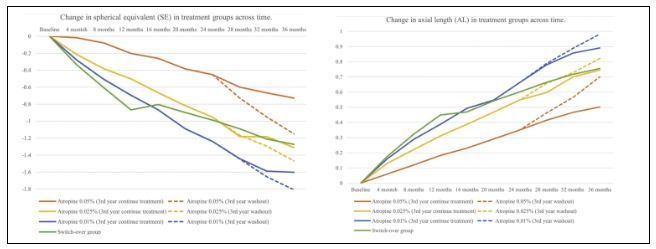
Compared with the sub groups of continuing treatment and stopping treatment in a single concentration group, the former had less myopia progression and axial growth. The difference in myopia progression between the two groups depends on atropine concentration and age: the lower the treatment concentration, the older the subject, and the smaller the difference in myopia progression between the two groups.
Therefore, compared with the withdrawal treatment, the myopia degree and ocular axis of children who continue to use low concentration atropine in the third year increase more slowly. It is recommended to continue to use atropine in the third year after 2 years of continuous low concentration atropine treatment.
2. The third stage of the lamp study showed that compared with 0.01% atropine, 0.05% atropine could bring better efficacy in the three-year continuous treatment regimen and stop regimen, and the efficacy of 0.05% atropine was more than twice that of 0.01%.
The average myopia progression of 0.05% atropine for 3 years was 0.73d, lower than 1.31d and 1.60d in 0.025% and 0.01% groups, and lower than the natural progression in the first year of placebo group (0.81d); The increase of ocular axis observed in 0.05%, 0.025% and 0.01% groups within 3 years were 0.40mm, 0.74mm and 0.89mm, respectively.
In the withdrawal group, the 3-year cumulative myopia growth of 0.05%, 0.025% and 0.01% atropine was 1.15d, 1.47d and 1.81d respectively, and the 3-year axial growth was 0.70mm, 0.82mm and 0.98mm.
After 3 years, the control effect of atropine at 0.05% concentration on myopia and ocular axis was more significant than that at other low concentrations, whether in the continuous treatment group or the withdrawal group.
3. In the 0.05%, 0.025% and 0.01% atropine groups, 0.05% atropine proved to be the most effective concentration in 3 years.
The rebound effects of 0.05%, 0.025% and 0.01% atropine groups were -0.68d, - 0.57d and -0.56d, respectively, following a concentration dependent response, but with a smaller amplitude. The difference between different concentration groups was too small and had no significant clinical and statistical significance. Therefore, considering the rebound effect of different concentrations, 0.05% atropine proved to be the most effective concentration in 3 years in the atropine groups of 0.05%, 0.025% and 0.01%.
4. Age also affects the rebound amplitude. The older the subject is, the smaller the rebound effect is.
In the third year, older age and lower concentrations of atropine after withdrawal were associated with myopia progression and decreased rebound of increased ocular axis. For each 1-year increase in the subject's age, myopia rebound decreased by 0.08d and Al elongation decreased by 0.05mm, while myopia rebound decreased by 0.20d and axial elongation decreased by 0.08mm in children with 0.01% atropine compared with 0.05% atropine. The difference in rebound amplitude between atropine groups of 0.05%, 0.025% and 0.01% decreased with age.
For younger children (6 to 8 years old), the myopia rebound after atropine withdrawal at the concentration of 0.05% is greater than that at the concentration of 0.01%; For the other two older age groups, the rebound amplitude of myopia related to the three concentrations was similar.
It is suggested to adopt the "weaning strategy" from high concentration to low concentration, and stop the treatment when the progress of physiological myopia slows down and the rebound effect is less.
5. During the 3-year period, the clinical tolerance of each low concentration atropine was good.
After stopping treatment in the third year, the pupil size and adaptive amplitude of children in each atropine concentration subgroup after stopping treatment returned to the baseline level. In the 0.05%, 0.025% and 0.01% atropine subgroups that continued treatment, the change of adaptive amplitude by the end of the third year was similar to the concentration dependent response at the end of the previous two years. Similarly, by the end of the third year, the change of pupil size was similar to the concentration dependent response at the end of the previous two years. In all subgroups, long-distance BCVA and short-distance BCVA were not affected.
In the third year, the number of subjects with photophobia remained similar in the two groups. The incidence of allergic conjunctivitis was similar in all concentration groups. Twenty subjects reported serious adverse events requiring hospitalization, but none of them were related to external atropine treatment. No differences in vision related quality of life were observed between all concentration groups and subgroups.
The results of phase 3 of the lamp study showed that even considering the rebound phenomenon of each concentration group, 0.05% concentration was the best concentration to control myopia in Chinese children in three years. In the third year, continued use of 0.05% atropine was better than stopping treatment. The greater rebound effect was associated with higher treatment concentrations and younger age at the time of discontinuation of treatment.
All concentrations of atropine were well tolerated and had no significant adverse effects on vision related quality of life. Considering that the efficacy of 0.05% atropine in 3 years is more than twice that of 0.01% atropine, and there is only a slight rebound effect, the study recommends that the treatment of 0.05% atropine in Asian children should continue until the third year.
References:
yam JC, Zhang XJ, Zhang y, et al. Three year clinical trial of low concentration atropine for myopia progress (lamp) study: continued versus washout: phase 3 report Ophthalmology. 2022 Mar; 129(3):308-321. doi: 10.1016/j.ophtha. 2021.10.002. Epub 2021 Oct 7. PMID: 34627809.