Hifn protects against nerve damage caused by ocular contusion by activating trkb
Shweta modgil from Emory University shared the research results of neuroprotection during ocular contusion. The team found that trauma will damage the optic nerve and brain vision center, which is manifested by functional vision loss, blurred vision / diplopia, dyslexia and photophobia. It is most often seen among military personnel performing combat missions and traffic accident survivors. Wearing head protectors and goggles can reduce the incidence of eye penetrating injury, but the injury caused by closed eye injury (ocular contusion) cannot be avoided.
Studies have found that trauma can lead to eye inflammation, oxidative stress, reduce the survival rate of RGC cells, cause nerve damage, and then lead to visual loss. After injection of BDNF into the mouse trauma model, it can combine with TrkB receptor to activate the downstream plc/akt/erk signal, and then reverse this damage to a certain extent, so as to achieve the purpose of treatment. However, there are some defects in the treatment strategy of BDNF. BDNF has a short half-life and cannot cross the blood retinal barrier, which brings difficulties to its transformation and application.
NAS is an endogenous TrkB receptor with a half-life of 30 minutes; Hioc is a derivative of NAS and has the effect of activating TrkB. It can cross the blood retinal barrier and act in the retina for up to 6 hours. Hioc injection can prevent vision loss caused by trauma and maintain the thickness of RGC nerve fiber layer.
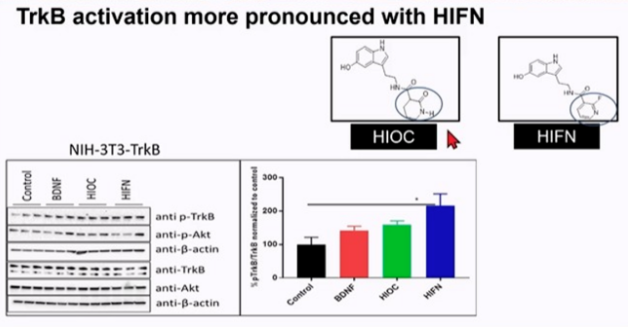
In vitro experiments showed that the addition of hioc analogues to primary ganglion cell cultures and nih-3t3-trkb cell lines could activate TrkB receptors. Hifn can achieve more obvious activation effect than hioc (P < 0.05).
Further research found that Hifn can effectively protect RGC cells and reduce visual loss in the mouse explosion injury model.
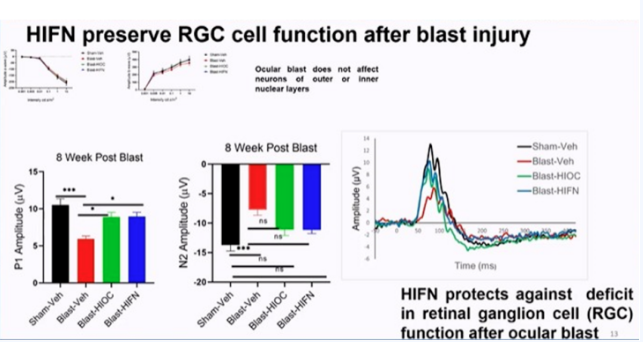
By inhibiting the activation of TrkB by Hifn, the neuroprotective and visual maintenance effects of Hifn were significantly reduced.
Summary: blast injury can lead to decreased contrast sensitivity, vision and RGC function; Hifn activated TrkB receptor has a protective effect on the decline of contrast sensitivity and visual acuity caused by explosive injury, and can protect the function of RGC cells.
Baicalin alleviates microglia mediated neuroinflammation and neurodegeneration in mouse retina
Li pan from Hong Kong Polytechnic University introduced the relevant mechanisms of retinal ischemia in eye diseases, including neurodegeneration caused by retinal blood flow obstruction; It occurs in retinal artery / vein occlusion, diabetes retinopathy (diabetes macular ischemia), glaucoma, etc., resulting in visual loss and blindness; And uncontrolled neuroinflammation, characterized by the activation of immune cells and the production of inflammatory factors, is an important cause of many neurodegenerative diseases.
Baicalin (BA) is a kind of flavonoids extracted from Scutellaria baicalensis Georgi. It has many effects, such as anti-inflammatory, anti-oxidation, anti apoptosis and anti-virus. Studies have shown that in PD and ad animal models, Ba treatment can effectively improve learning and memory function by inhibiting inflammation and metabolic dysfunction caused by astrocytes; In the rat model of cerebral ischemia / reperfusion injury, oral administration of Ba can regulate neuroinflammation and neurobehavioral defects caused by microglia / macrophage activation; In diabetes retinopathy rats, oral administration of Ba can significantly inhibit the over activation of retinal immune response and the expression of proinflammatory cytokines. Therefore, researchers used lipopolysaccharide (LPS) to induce BV2 microglia activation, and Ba intervention significantly inhibited the expression of proinflammatory factors; In vivo experiments showed that Ba could improve the morphological changes of microglia in retinal ischemia-reperfusion (i/r) mouse model, and inhibit the expression of activated microglia markers and proinflammatory cytokines; Prevent RGC cell degeneration caused by ischemia-reperfusion (i/r); Protect retinal physiology and visual function.
Summary: both in vivo and in vitro experiments have proved that Ba is a negative regulator of activated microglia and immune response in the ischemia-reperfusion (i/r) mouse model, which can effectively alleviate nerve injury and visual function damage.
K-ras V12 can promote RGC cell regeneration after optic nerve injury!
Researchers from Tokyo Metropolitan Medical Research Institute explained the mechanism why the optic nerve cannot regenerate: once the optic nerve is damaged, it will cause glial cell scar, while the innate regeneration ability of neurons in the central nervous system is limited. Many studies have shown the mechanism of this congenital limited regenerative ability, including the activation of BDNF downstream, which leads to the activation of pi3k/akt/mtor pathway and ras/raf/erk1/2 pathway, thus playing a role in neuroprotection and axonal extension.
Ras protein is a member of a large family of small GTP binding proteins with GTP hydrolase activity. The conformation of Ras protein circulates between two different conformational states: when Ras protein binds GTP, it is in an active state; When Ras protein binds to GDP, it is inactive. Researchers focused on ras by constructing a constitutive active mutant K-ras V12, a GTP hydrolase inhibitor. The COS-7 cell line was used to verify the function of K-ras V12 in activating pi3k/akt/mtor pathway and ras/raf/erk1/2. The results showed that K-ras V12 could enhance the phosphorylation level of Akt and ERK.
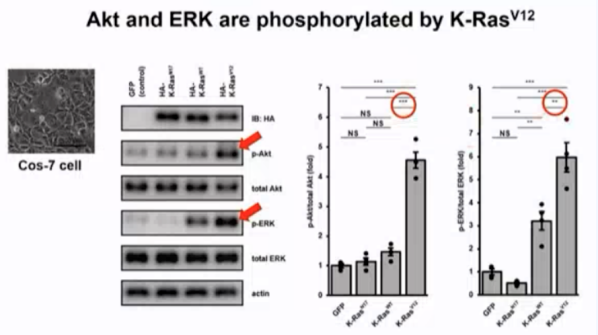
Based on the conclusion that K-ras V12 can effectively enhance the phosphorylation level of Akt and ERK in the above in vitro experiments, the researchers decided to verify whether K-ras V12 has neuroprotective effect in vivo. Optical nerve crush (ONC) optic nerve injury model is used, which can cause retinal RGC cell death. Aav-k-ras V12 was injected into vitreous cavity and transfected into retinal RGC cells. The results showed that the experimental group of mice could effectively protect the density of RGC cells after optic nerve injury compared with the blank control group. This protective effect was verified again in vitro.
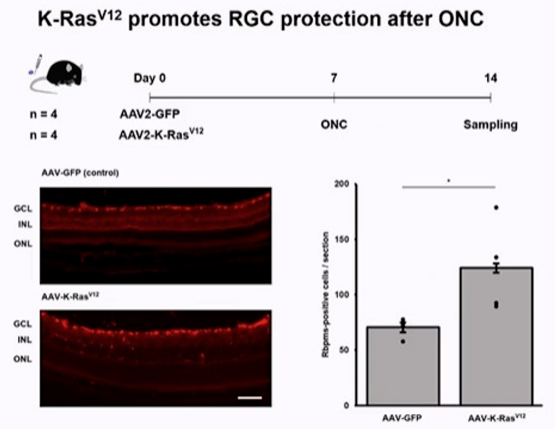
Through further research, the researchers verified that k-rasv12 in vivo did not cause RGC cell tumorigenesis and continuous proliferation, and the proliferating cells were microglia.
Summary: k-rasv12 effectively activates pi3k/akt and raf/mek/erk signal cascades in vitro. K-rasv12 promotes RGC protection after onc and has no obvious tumorigenic effect on RGC.
Inhibiting DNA methylation can effectively protect RGC cells during the progression of glaucoma
Shahid Husain, from the ophthalmology department of South Carolina Medical University, listed the risk factors of glaucoma, including high intraocular pressure, glial cell activation, production of reactive oxygen species, neuroinflammation, epigenetic changes (histone modification and DNA methylation), and described the relationship between DNA methylation and glaucoma. It is reported that the promoter region methylation of prss50 gene is significantly increased in whole blood samples of retina and AMD patients. However, there is no genome-wide methylation database of retina, optic nerve or intraocular cells in glaucoma patients. Limited studies in the anterior segment showed that dotted global DNA methylation of the HERV-K repeat was observed at the differentially methylated CpG site in the outflow resistance pathway of trabeculectomy specimens, trabecular meshwork cells and Schlemm duct cells in POAG patients.
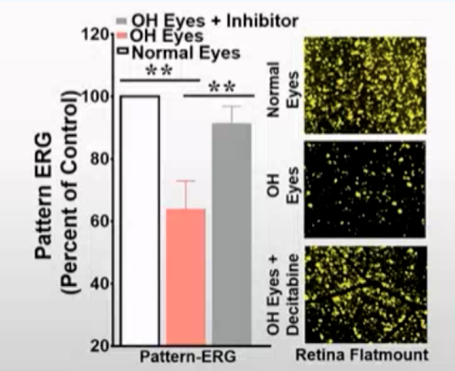
High intraocular pressure may lead to increased DNA methylation, which may lead to neurotrophic problems, and ultimately lead to a decline in the survival rate of RGC cells; Methylation inhibitors are expected to reduce this reaction and ultimately improve the survival rate of RGC cells. Based on this, researchers conducted a series of studies, using DNA methylation inhibitors to intervene in the animal model of high intraocular pressure (oh model), and found that the inhibitor intervention group can effectively improve the survival rate of RGC cells.
The detection of methyltransferase 1 and 3a (dnmt1&dnmt3a) in normal group, pure high intraocular pressure OH group and high intraocular pressure Oh application inhibitor group showed that the expression of DNMTs protein was significantly increased in pure OH group at 7 and 42 days after the modeling of high intraocular pressure model, while the expression of DNMTs decreased after the use of inhibitors.
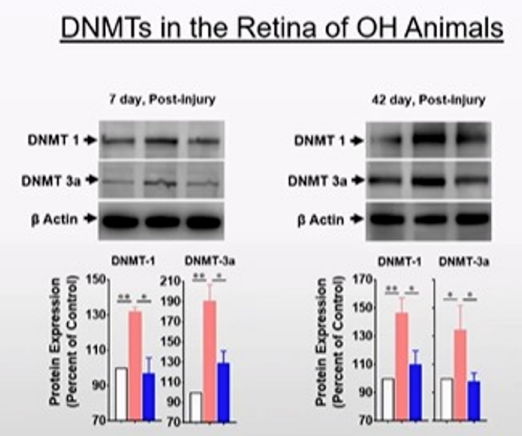
Further studies found that methylation inhibitors can inhibit the expression of Dnmt3a and DNMT3b in retinal RGC cells and optic nerve in the model of high intraocular pressure.
Summary: DNA methylation plays an important role in the mechanism of RGC cell death in glaucoma ocular hypertension model; RGC cells can be effectively protected by inhibiting DNA methylation.
Endothelin receptor antagonist maxitetan macitentan promotes neuroprotection of retinal ganglion cells through vascular and cellular mechanisms
Ragnu Krishnamoorthy from the North Texas Institute of Ophthalmology (nteri) introduced the mechanism of ET-1-mediated neurodegeneration in glaucoma: various pathogenesis of glaucoma, including vascular abnormalities, high intraocular pressure, neuromodulation, RPE layer abnormalities, etc., will stimulate the production of ET-1, which will lead to nerve related axonal conduction abnormalities, optic astrocyte proliferation, hypoxia / ischemia, and increased neurotoxicity, It leads to oxidative stress and changes in extracellular matrix, and finally causes RGC apoptosis and optic nerve damage, which is also an important mechanism of glaucoma and other neuropathy. Therefore, researchers hypothesized that the use of endothelin receptor antagonists may promote the neuroprotective process of glaucoma through vascular and intracellular mechanisms.
First, the endothelin receptor antagonist macitentan/et-1 was used for intravitreal injection. After ET-1 injection, retinal vessels became sparse, while the retinal vessel density and vessel diameter of macitentan+et-1 group were higher, indicating that endothelin antagonist can antagonize the vascular injury of ET-1.
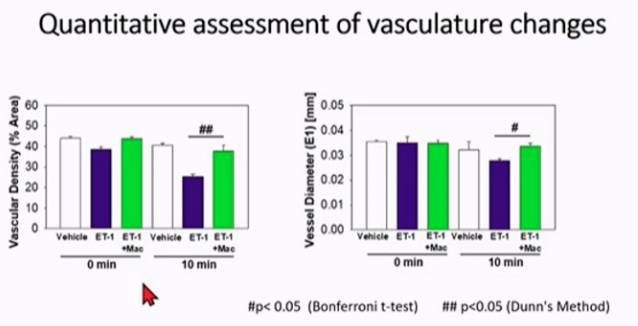
Further analysis showed that the application of endothelin receptor antagonist macitentan in the eyes of rats injected with ET-1 could significantly reduce the loss of RGC cells. The use of macitentan in the high intraocular pressure model can effectively improve the survival rate of RGC cells under high intraocular pressure stimulation after 4 weeks, play a neuroprotective role, and the optic nerve injury is also reduced in this process. ERG was used to analyze the function of RGC cells. It was found that the function of RGC cells could be effectively improved after macitentan was applied in the high intraocular pressure model for 2 weeks, and the effect lasted until 4 weeks after treatment. It shows that endothelin receptor antagonist has the effect of protecting RGC function in glaucoma model.
Summary: oral endothelin receptor antagonist macitentan can reduce the fundus vascular injury and RGC cell loss caused by intravitreal injection of ET-1; Oral endothelin receptor antagonist maxitetan macitentan can alleviate RGC cell loss and optic nerve injury in high intraocular pressure model; Oral endothelin receptor antagonist maxitetan macitentan can be used as a potential therapeutic drug for neuroprotection in the treatment of glaucoma, which provides a new idea for clinical treatment.