Macular dystrophy: a typical feature of sorsby fundus dystrophy
Macular dystrophy includes heterogeneous diseases characterized by central visual loss, including ABCA4, best1, Prph2, TIMP3 and other pathogenic gene mutations. Pathogenic mutations in TIMP3 are associated with sorsby fundus dystrophy (SFD), which is typically characterized by the presence of vitreous warts and their accumulation in the subretinal pigment epithelium space. SFD usually starts in adults. Patients have night blindness symptoms, choroidal neovascularization, and secondary acute visual changes, which is a common feature of the late stage of the disease. TIMP3 gene encodes tissue inhibitor of metalloproteinase-3, which is secreted by retinal pigment epithelium and enters Bruch membrane, a multilayer extracellular matrix (ECM) that separates retinal pigment epithelium from choroidal capillaries. Previously, SFD related gene mutations were all located in the TIMP3 mature protein sequence (Figure 1). Most of these variants exist in the C-terminal domain and introduce additional cysteine, which may be conducive to intermolecular dimerization and lead to the accumulation of variant proteins in ECM. This is the main mechanism theory of SFD pathobiology. Recently, researchers found two families of macular dystrophy associated with TIMP3 signal peptide variants, which lack typical SFD characteristics.
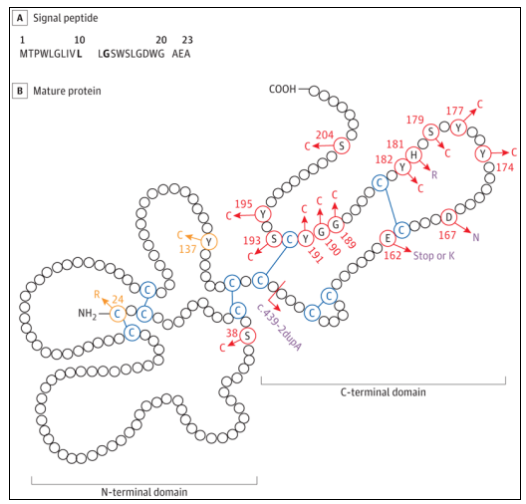
Figure 1 TIMP3 mature protein structure
Analysis of special types of patients: clinical imaging and molecular genetic testing
This case series was composed of two families with early-onset diffuse maculopathy. The correlation analysis was carried out at the National Institute of Ophthalmology, clinical center of the National Institutes of health. The data were collected and analyzed from October 2009 to December 2021. Researchers conducted clinical imaging and molecular genetic tests on two families with macular dystrophy. Co segregation analysis of TIMP3 variants was performed in affected and unaffected family members. Cleavage defects of candidate TIMP3 signal peptide variants were evaluated after transfection.
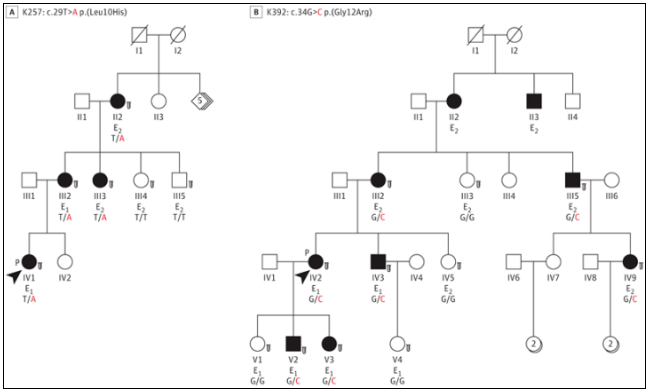
Figure 2 Macular dystrophy pedigree with TIMP3 signal peptide variation
It is found that patients have different genotypes: it may lead to different disease manifestations
Researchers identified two families of autosomal dominant macular dystrophy and selected individuals from families K257 (n=2) and k392 (n=4) for detailed clinical studies. The common feature of all affected individuals in these two families (the disease first started in adolescence to before the age of 40) is the presence of diffuse macular disease. The results show that it is mainly paracentral atrophy that leads to paracentral scotoma.
In the K257 family, cystic changes affecting central vision were observed in the originator of the disease (individual iv1). Fluorescence fundus imaging showed mottled low autofluorescence, and young patients showed high autofluorescence accumulation. Optical coherence tomography is of great significance to the patchy extraretinal and retinal pigment epithelium atrophy areas and extensive extraretinal degeneration areas in the whole macula.
In the k392 family, the originator of the disease (individual iv2) developed in adulthood, and the examination of their children found that the macular lesions did not shrink significantly. It is worth noting that the useful central vision of the affected individuals was preserved, and no choroidal neovascularization occurred, including 2 patients with retinopathy for more than 20 years.
Researchers then evaluated the molecules of retinal dystrophy related genes and found that heterozygous mutations were located in the TIMP3 signal peptide sequence, including c.29t>a P. (leu10his) in K257 family and c.34g>c P. (gly12arg) in k392 family (Fig. 3). All individuals assessed to have specific disease manifestations (Figure 2; 6 individuals in the K257 family and 11 individuals in the k392 family) have phenotypic associations. Previously reported SFD related mutations are located in mature protein sequences, most of which are located in the C-terminal domain. Considering that individuals with mutations in TIMP3 signal peptide do not have bleeding or neovascularization in typical SFD, it is assumed that these two variants lead to TIMP3 related retinopathy by changing protein transport, and there are different disease mechanisms.
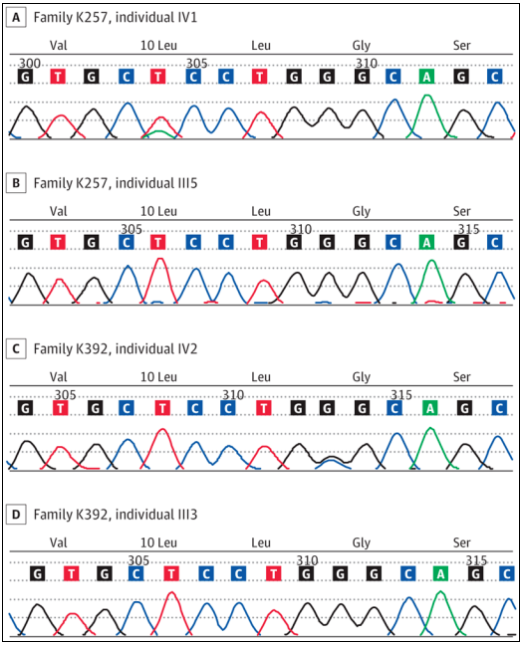
Figure 3 Gene sequencing results
Analysis of protein cleavage products: different gene variants have different protein cleavage transport processes
Although all mutant individuals showed impaired ECM deposition, the researchers hypothesized that the mechanism of l10h and g12h was defective signal peptide cleavage, rather than TIMP3 aggregation observed in other SFD related variants. Therefore, the researchers further detected specific TIMP3 species in different cells. Five wild-type bands, including dimer, precursor, glycosylation, non glycosylation and degradation products, were found in the soluble extracts of cytoplasm, cell membrane and nucleus. L10h variants are mostly non cleared precursors in cytoplasm, nucleus and cell membrane group, and slower bands are observed in membrane and nuclear extracts, which may represent glycosylated species. Typical SFD related mutations show a normal cleavage pattern. The data showed that l10h and g12r signal peptide mutations affected the cleavage and transport of proteins, thereby reducing the mature TIMP3 protein in ECM.
The researchers analyzed the TIMP3 variant (l10h or g12r) of 11 individuals from two families of early-onset diffuse maculopathy without choroidal neovascularization, and confirmed the co segregation of genotype and phenotype in other unaffected family members. Biochemical analysis confirmed the defects of protein maturation and extracellular deposition. This study found that TIMP3 variants that alter the function of signal peptides deviate from the classic sorsby fundus dystrophy in phenotypic characteristics and potential mechanisms. These results suggest that atypical patient manifestations are caused by defects in TIMP3 signal peptide, which is related to impaired protein division and deposition into extracellular matrix, and involves a new macular dystrophy disease.
References: Bin Guan, PhD, laryssa a. huryn, MD, Andrew B. Hughes, BS, Zhiyu Li, MD, Chelsea bender, BS, Delphine blain, MS, MBA, Amy turiff, MS, Cathy a. cukras, MD, PhD, Robert B. Hufnagel, MD, phd Early-onset TIMP3-related retinopathy associated with impaired signal peptide. doi:10.1001/jamaophthalmol. two thousand and twenty-two point one eight two two