Source: Chinese Journal of laboratory medicine, 2022,45 (1): 14-23
abstract
Infectious eye disease is one of the important causes of blindness in developing countries. Pathogen examination is the gold standard for the diagnosis of infectious eye disease. Due to the difficulty in obtaining ocular tissues and the small amount of samples, the positive rate of laboratory diagnosis is low, which seriously affects the diagnosis and prognosis of the disease. The laboratory diagnosis ability of infectious eye disease in most medical institutions in China is insufficient, and there is a lack of standardized methods for ocular microbial detection. Therefore, the laboratory branch of Beijing Medical Association organized experts and relevant research groups to summarize the pathogenic diagnosis path suitable for the clinical practice of ophthalmology in China through discussion and clinical verification, so as to form the consensus of laboratory diagnosis experts of infectious ophthalmopathy. The consensus starts with the clinical manifestations and inspection indications of common infectious eye diseases, and standardizes the methods and processes of eye specimen collection, transportation and quality evaluation; It is emphasized that ophthalmologists should fully cooperate with microbiological inspectors, correctly select appropriate microbiological detection methods, advocate bedside inoculation, and put forward suggestions on scraping cytology, traditional microbial culture, Acanthamoeba culture, nucleic acid detection, metagenomic sequencing and other technologies.
Infectious ophthalmopathy is a kind of blinding disease caused by pathogenic microorganisms infecting eye tissue and its appendages (conjunctiva, cornea, anterior chamber, vitreous body, eyelid margin and lacrimal apparatus, etc.), resulting in local tissue damage, functional damage and visual loss [1]. Due to the limitations of health conditions and medical level, this kind of disease has always been a high incidence in low - and middle-income countries. The incidence rate of infectious keratitis in China is 5 times that of developed countries (about 0.04%), reaching 0.192% (1, 2, 3). The incidence of endophthalmitis is also increasing. The incidence rate of infectious endophthalmitis after cataract surgery is 0.11% (4), almost 10 times that of developed countries (0.012%~0.053%) [5, 6, 7, 8]. However, most medical institutions in China lack the ability of microbial detection of infectious eye diseases, and lack of clinical experience with reliable etiological evidence. In 2018, the Inspection Branch of Beijing Medical Association organized relevant experts to hold a special discussion on this issue, learn from the advanced ophthalmic etiology inspection methods at home and abroad, and establish a standardized method suitable for general hospitals to carry out this work. On this basis, the laboratory branch of Beijing Medical Association entrusted Beijing Institute of Ophthalmology and Beijing Tongren Hospital Affiliated to Capital Medical University to carry out relevant clinical verification. After three years of efforts, after analyzing and sorting out the diagnosis and treatment data of 1256 cases of different types of infectious eye diseases, we summarized the path, technical means and application scenarios suitable for ophthalmic etiology diagnosis in China, and formed the following expert consensus.
1、 Common pathogens and inspection indications
Ophthalmic clinical examination and microbiological laboratory examination are two indispensable parts of the diagnosis of infectious ophthalmopathy. Therefore, in the process of laboratory diagnosis, the laboratory physician needs to clearly understand the source of samples and the clinical manifestations of patients, so as to make a preliminary judgment on the possible pathogens in various parts of the eye and the microorganisms that may be related to different types of eye infection. To this end, this consensus first briefly introduces the anatomy of the eye (Fig. 1), so that the examiner can clarify the location of materials and the source of specimens on the basis of understanding the anatomy of the eye; Secondly, the examination indications and specific examination contents of common infectious eye diseases are introduced in detail (Table 1), and the pathogenic diagnosis path is established (Fig. 2).
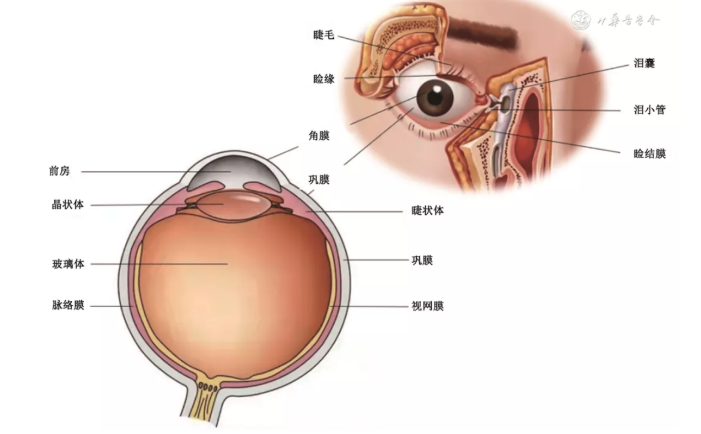
Figure 1 anatomical diagram of eye and surrounding appendages
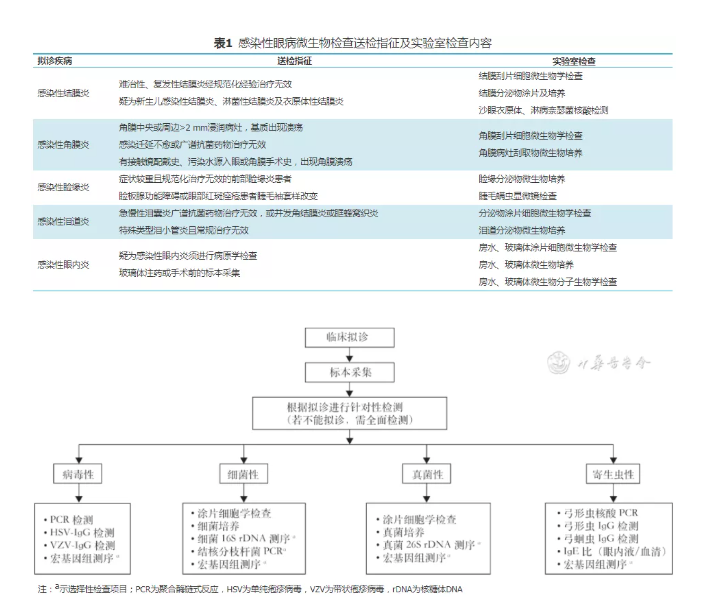
Figure 2 pathogenic diagnosis path of infectious ophthalmopathy
1. Infectious conjunctivitis: infectious conjunctivitis is easy to occur when the conjunctiva is exposed to the external environment. Conjunctival congestion and increased secretion are the typical clinical features. Most conjunctivitis do not need microbiological examination. However, conjunctival scraping, culture or nucleic acid detection are of great significance for the diagnosis and treatment of epidemic, refractory, recurrent or ineffective infectious conjunctivitis [9]. The common pathogens of infectious conjunctivitis are Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae and others α- Hemolytic streptococcus, Enterobacteriaceae, Acinetobacter, Corynebacterium, adenovirus (children type 3 and 7; adults type 8, 11 and 19), herpes simplex virus (types 1 and 2), Coxsackie virus a 24 and enterovirus 70, etc. Chlamydia trachomatis serotype A ~ C can cause trachomatic conjunctivitis, and Chlamydia trachomatis serotype D ~ k can cause inclusion body conjunctivitis [10,11].
2. Infectious keratitis: infectious keratitis is mostly caused by the invasion of pathogenic microorganisms into the cornea. Those with suppurative keratitis (bacterial, fungal and Amoeba infection) need to be diagnosed through laboratory examination [12]. For patients with non suppurative (viral) keratitis, the clinical diagnosis can be based on the history of recurrent episodes and the characteristics of corneal lesions. Due to the small amount of materials taken from corneal lesions, the specimens should be immediately smeared, stained and microscopic examined or inoculated with microorganisms beside the bed; In addition to routine corneal scraping and culture, pathogen nucleic acid detection can also be selected. Bacterial keratitis is mainly infected by Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pneumoniae, hemolytic streptococcus, Pseudomonas aeruginosa (contact lenses) and Enterobacteriaceae [13]; Atypical mycobacteria (Mycobacterium Chelonii, incidental Mycobacterium, Mycobacterium abscess) are rare, and are generally found in patients after trauma or refractive surgery. Viral keratitis is mainly caused by herpes simplex virus (types 1 and 2), varicella zoster virus, Coxsackie virus A24 and enterovirus 70. In addition, Ebola virus and arbovirus infection (Zika virus, dengue fever and Chikungunya virus) can cause mild keratitis [14]. Fungal keratitis often occurs in patients with plant trauma or chronic ocular surface diseases. The common filamentous fungi are Fusarium, Aspergillus, hyphomycetes, Paecilomyces, Cladosporium and Curvularia. Keratitis caused by Candida and Cryptococcus can occur in patients with low immune function [15]. Parasitic keratitis is rare, mainly caused by Acanthamoeba protozoa. In addition, there is keratoconjunctivitis caused by microsporidia, amoeba and fly maggot larvae [16].
3. Infectious blepharitis: if the blepharitis appears as one of the lower body signs, it can be suspected as infectious blepharitis. (1) Hyperemia at the palpebral margin, eyelashes falling off, and small pustules, scales or cuff like substances can be seen at the base of eyelashes; (2) For patients with anterior blepharitis whose empirical treatment is ineffective, blepharitis secretion culture can be carried out [17] (aerobic and anaerobic culture can be carried out at the same time); (3) For patients with meibomian gland dysfunction or ocular erythema acne, when there are cuff like changes in eyelashes, eyelash mite microscopic examination is required. Smear and culture are helpful to find pathogens. Common pathogens include Staphylococcus aureus, Staphylococcus epidermidis, dermatobacterium acnes, yeast (Bacillus scurf), hair follicle Demodex (eyelash root), fly maggot larvae and pubic lice. Pay attention to distinguish normal flora [18].
4. Infective dacryocystitis: the drainage system of tear is called lacrimal duct. If the lower body sign appears, it can be diagnosed as lacrimal duct infection, such as lacrimal overflow, viscous secretion of inner canthus, redness and swelling of lacrimal dots or lacrimal sac, and purulent secretion overflow can be seen after squeezing lacrimal dots or lacrimal sac. Patients with lacrimal canaliculitis or dacryocystitis complicated with keratoconjunctivitis or orbital cellulitis who fail to respond to empirical treatment need to undergo cell microbiological examination of lacrimal passage secretions [19]. Common pathogens include actinomycetes (actinomycetes and Nocardia are the most common), Streptococcus pneumoniae, hemolytic streptococcus, Haemophilus influenzae, Staphylococcus aureus and gram-negative bacilli. Lacrimal duct fungi (aspergillus, Alternaria alternata, Candida, Cladosporium) infection is rare [20].
5. Infective endophthalmitis: it refers to the infection caused by microorganisms in the eye. It is a clinical acute and severe blinding eye disease, including endophthalmitis (bacteria, fungi), viral endophthalmitis, parasitic endophthalmitis, etc. Infectious endophthalmitis is frequently associated with open ocular trauma, history of intraocular surgery, diabetes, organ transplantation, and long term use of immunosuppressive agents. It is characterized by redness, eye pain, tears, decreased vision, ciliary congestion, aqueous humor flash, aqueous humor floating cells and even anterior chamber pus, posterior pupillary adhesion, obvious vitreous opacity, pre retinal pomponema, etc. [21]. Exogenous endophthalmitis is common in postoperative infection and ocular trauma (especially after cataract and glaucoma surgery). Common pathogens include Staphylococcus, Enterococcus, Streptococcus, enterobacter, non fermenting bacteria, aerobic bacillus, Aspergillus, Fusarium, dark fungi, etc. it is characterized by mixed infection of multiple pathogens, among which coagulase negative staphylococcus is easy to cause acute infection, Dermatobacterium acnes is the most common cause of chronic infection. Be alert to the ocular infection of highly virulent Klebsiella pneumoniae. Pseudomonas and filamentous fungi are rare [22]. Endogenous endophthalmitis mostly comes from blood borne infection. The common pathogens are Streptococcus pneumoniae, Staphylococcus α- Hemolytic streptococcus, Mycobacterium, yeast, filamentous fungi, Enterococcus, anatrophic cocci, Streptococcus granulosus, Haemophilus influenzae, veronicus, Prevotella, Candida, etc. Retinitis is mainly caused by virus infection, including herpes simplex virus, cytomegalovirus, herpes zoster virus, EB virus, etc. [23]. Inflammation of iris, ciliary body and / or choroid can be related to systemic or local infection from bacterial sources (Borrelia burgdorferi, Treponema pallidum, Mycobacterium, Leptospira, Bartonella), viral sources (herpesvirus, cytomegalovirus) or parasitic sources (Ascaris lumbricoides, Toxoplasma gondii, onchocercaria, cysticercus cellulosae). Toxoplasma gondii is the culprit of chorioretinitis and the main cause of infectious posterior uveitis [24].
2、 Specimen collection and quality evaluation
(1) Principles of specimen collection and transportation
The following rules are formulated with reference to the 2018 U.S. microbiological laboratory operation guide, the 2021 U.S. clinical microbiological laboratory practical guide for ocular infection and the 2021 European Microbiological manual [25, 26].
1. Emphasize the principle of "immediate submission for examination, immediate smear and bedside inoculation", especially the specimens requiring anaerobic culture; If it is difficult to achieve immediate inoculation, it shall be stored at room temperature and the collection time shall be marked. The methods of specimen collection, storage and transportation are shown in Table 2.

2. In principle, the specimens used for culture shall be examined for cell microbiology at the same time; For PCR specimens, the use of talc, sodium fluorescein and eye anesthetics should be avoided.
3. As eye samples are precious and small in quantity, the doctor should inform the laboratory of specific test items and priorities.
4. Conjunctival secretions should be taken from both eyes and the eye type and location should be indicated in the application form, so as to facilitate the analysis of normal flora of conjunctival sac.
5. Corneal ulcer specimens should be gently scraped by an ophthalmologist or ophthalmologist with clinical experience under a slit lamp microscope with a sterile spatula or sterile surgical blade, smeared immediately and inoculated beside the bed. Corneal tissue and biopsy specimens are recommended to be cut with sterile scissors or surgical blade before inoculation; Intraocular specimens (aqueous humor or vitreous body) need to be taken surgically and smeared and inoculated with stock solution.
6. Intraocular fluid includes aqueous humor and vitreous humor. Invasive intraocular fluid detection is only applicable to patients with highly suspected infectious endophthalmitis. It must be collected by experienced ophthalmologists in a sterile environment. Antibiotics (such as ofloxacin and tobramycin) eye drops are needed before operation. After surface anesthesia, the skin is disinfected and povidone iodine conjunctival sac is rinsed. The appropriate amount of aqueous humor should be 0.05 ~ 0.1 ml, and the vitreous lavage fluid should be centrifuged (800 RPM) × After 10 min, centrifugation radius 10 cm), take the precipitation smear or shake the smear for microscopic examination or microbial culture. 0.22 can also be used μ M filter with sterile filter and culture the filter membrane with bacteria and fungi. Aqueous humor or vitreous specimens can be injected into a sterile EP tube for transfer, or the syringe, together with the needle and needle cap, can be placed in a closed specimen transfer box for rapid transfer.
7. The foreign body can be cultured according to the condition of the eye, but the results of contact lens detection can also be interpreted with caution.
(2) Specimen quality assessment
Eye samples are not recommended to be rejected. Even if the number of samples is small and the transfer is overtime, the inspection should be completed as far as possible, and the clinician should be communicated in time to strive for the second inspection. The evaluation of specimens should follow the following principles.
1. Evaluation of scraping specimens: specimens for microbiological examination of scraping cells, such as secretion, pus or necrotic tissue, shall be made into uniform thin-layer smear with a diameter of about 10 mm. Except that the vitreous lavage solution needs to be centrifuged and precipitated or tossed, the rest should use the original specimen. (1) Evaluation of tissue specimens: secretions, pus or necrotic tissue can be seen on the slide with the naked eye, and a thin layer is tiled without tissue cell accumulation. (2) Corneal scraping evaluation: the number of exfoliated or inflammatory cells in and around the keratoconjunctival lesions must be ≥ 1. Internationally, the number of scraping cells (all cells visible under the microscope) is divided into four levels, that is, the cell count under the low power microscope (LP) shall prevail, level 0: ≤ 10 / LP, level 1: 11 ~ 50 / LP, level 2: 51 ~ 100 / LP, Level 3: 101 ~ 1000 / LP, and level 4: > 1000 / LP [27]. Other tissue specimens (such as aqueous humor and vitreous) can also refer to this standard.
2. Evaluation of cultured samples: including whether to use antibiotics before sample collection, type and time of antibiotics; Collection mode, transfer device and sampling quantity; Whether it is inoculated beside the bed and delivered to the laboratory within 2 hours; Apply for the suitability of inspection items, etc. Since it is not recommended to reject specimens, specimens that fail to pass the evaluation need to be described in the result report.
3、 Laboratory examination
(1) Microbiological examination of scraping cells
For laboratory diagnosis of infectious ophthalmopathy, except for ciliary mites, pubic lice or conjunctival sucking nematodes, cell microbiological examination should be carried out before culture, nucleic acid and immunological detection, and attention should be paid to ocular scraping cytology from the perspective of pathology. The choice of scraping staining method depends on the clinical diagnosis and suspected pathogen type. Common dyeing methods include Giemsa dyeing, gram dyeing, fluorescence dyeing and acid fast dyeing. See Table 3 for common dyeing methods and inspection objects.
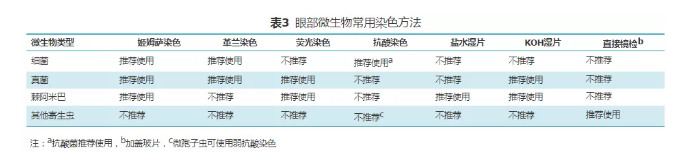
1. Giemsa staining: it is used for histopathological and pathogen morphological examination. It is the most commonly used staining method in ophthalmology. Bacteria, inclusion bodies, fungi, amoebas and other microorganisms can be identified according to the morphological characteristics and arrangement of pathogens. The microscopic characteristics of common pathogenic microorganisms in the eyes after Giemsa staining are shown in Table 4. Giemsa staining can also observe the corneal conjunctival epithelial cells, inflammatory exudative cells (neutrophils, lymphocytes, eosinophils, mononuclear phagocytes, plasma cells and mast cells) and special tissue cells at the lesion. Infection or non infection, acute infection (mainly neutrophils) or chronic infection (lymphocytosis) can be judged according to the proportion of inflammatory cells; At the same time, the types of inflammatory cells around different microbial infections are different, such as eosinophil infiltration in Acanthamoeba infection; The acute stage of Chlamydia infection is mainly neutrophil infiltration, while the virus infection is mainly lymphocyte infiltration, and intracellular inclusion bodies can appear.

2. Gram staining: microscopic description is the same as routine microbiological examination. Judge according to the bacterial morphology (bacilli, cocci and Coccobacteria), staining characteristics (positive, negative or uncertain), arrangement mode (in pairs, in piles or scattered, short chain or long chain, etc.), leukocyte phagocytosis and other factors. If a large number of neutrophils are seen in the smear, gram-positive diplococcus can be seen inside and outside the cell, which is arranged relatively in a spearhead like manner and has a thick capsule, it can be reported as suspected Streptococcus pneumoniae [28].
3. Fluorescence staining: the chitinase labeled with fluorescein can specifically combine with the polysaccharide and chitin of the fungal cell wall, and quickly judge whether the sample contains fungal components within 5 minutes. The tissue background is homogeneous black or weak blue fluorescence, while the fungal cell wall is bright blue purple or bright fluorescent blue, which is easy to identify the morphology, structure and mycelial density of hyphae or spores [29]. Because Acanthamoeba cysts also contain chitin, the cysts can also be light stained blue-green. Fluorescence staining can significantly improve the sensitivity of diagnosis of fungi, microsporidia and Acanthamoeba, but it should be noted that swab cotton fiber and any other substances containing cellulose or chitin may cause false positive.
4. Acid fast staining and weak acid fast staining: acid fast staining can detect eye acid fast bacteria, mainly including M. tuberculosis (MTB) and non tuberculosis mycobacteria (NTM). Weak acid fast staining can be used for suspected Nocardia infection or microsporidia, and it is positive if the bacteria or insects are pink.
5. Wet tablet method: normal saline wet tablet method and KOH wet tablet method are commonly used. The normal saline wet tablet method is mainly used for in vivo observation of Acanthamoeba trophozoites and cysts, especially for observing the movement of trophozoites. Acanthamoeba trophozoites migrate between corneal tissues, which are irregular and easy to change. Their cytoplasm is rich in granular material and can move with the deformation of trophozoites. The double wall of prepystal cyst has not been formed, and the cytoplasm contains uniform coarse particles, which are more active and vibrate constantly. Mature cysts have double-layer structure of inner and outer cyst walls. The empty sac is wrinkled and has no content, which is common after anti amoeba treatment. Koh wet slide method can also be used for the observation of filamentous fungi. Mix the collected samples and apply them on the clean glass slide dripping with 10% ~ 20% KOH solution, cover the glass slide, use the flame of alcohol lamp for 2 ~ 3 times, and observe with a low power microscope after cooling. Koh can digest tissue cells and make fungal hyphae clearly visible.
6. Parasite examination: in addition to Acanthamoeba protozoa, common eye parasites also include conjunctival sucking nematodes, eyelash mites and pubic lice. The conjunctival sucking nematode grows in the conjunctival sac. The white thread like worm can be seen under the slit lamp. Take out the tweezers and observe under the optical microscope. Mites are located in the ciliary follicles. Look for adults, larvae or eggs of Demodex under the light microscope (100 times). Pubic lice often grow in the root and stem of eyelashes. After cutting or pulling out eyelashes under slit lamp, adults and eggs are observed under optical microscope, which is easy to diagnose.
(2) Traditional microbial culture
1. Culture medium: routine culture of ocular microorganisms includes aerobic, facultative anaerobic, anaerobic and fungal culture, and micro aerobic and mycobacterial culture can be added if necessary. Common culture media include Columbia blood agar and chocolate agar. If necessary, add McConkey, sapaul, potato and anaerobic blood agar. Emphasize the importance of anaerobic culture. Meanwhile, ophthalmologists / technicians need to communicate closely with clinicians and carry out the test items conducive to diagnosis on the hard won samples as comprehensively as possible.
2. Culture conditions: the biochemical incubator is suitable for the culture of bacteria and fungi growing at different temperatures. Generally, the bacterial culture temperature is 34 ~ 36 ℃ for 2 ~ 5 days; The fungi were cultured at 27 ~ 29 ℃ or 34 ~ 36 ℃ for 7 ~ 30 days. The environment for bacterial culture includes aerobic and anaerobic (anaerobic bag, anaerobic tank and anaerobic glove box), 5% ~ 7% CO2 incubator, micro aerobic, etc; In addition to the conventional Columbia blood agar culture for 3 ~ 5 days, the growth of Mycobacterium requires Roche or Michaelis 7h10 medium and BACTEC MGIT culture system. The growth is observed after 7 days, and the longest culture time is 56 days. Ocular fungi are suitable for potato glucose agar medium or Sabouraud medium slope. Observe every day. The scraps / smears are positive, but the culture is negative. It is suspected that rare or slow-growing fungi should extend the observation period to 4 ~ 8 weeks.
3. Bacteria enrichment: bacteria enrichment is not recommended for eye infection samples, especially from secretions connected with the outside world, such as conjunctiva, lacrimal duct and lacrimal sac. Intraoperative aqueous humor, vitreous humor and cornea can be enriched.
4. Microbial identification: colony based biochemical test, automatic instrument and MALDI-TOF mass spectrometry are commonly used identification methods, which can realize rapid identification for both bacteria and fungi. When necessary, DNA sequencing (d118rs) or its (28D2) can be used for identification.
5. Antimicrobial susceptibility test: the standard of clinical and Laboratory Standards Institute (CLSI) [30] is basically selected in China, but ophthalmology is mostly used locally, and its local exposure dose is much higher than the serum drug concentration. The results of antimicrobial susceptibility test (AST) are only for reference. For external eye specimens, it is sometimes impossible to identify the isolated strains as symbiotic flora, contamination or infection with real pathogens. In addition to coagulase negative staphylococcus (non LUDEN Staphylococcus) and Corynebacterium diphtheriae, two dominant pathogens were pure cultured or cultured for a single microorganism, Identification and AST are required (Staphylococcus aureus and Pseudomonas aeruginosa should be identified and AST regardless of the number of colonies). In contrast, the inner eye specimen should be treated as a sterile site, and any culture positive pathogen needs to be identified and AST. If CLSI lacks the judgment standard of certain antibiotics, it can refer to the local epidemiological results.
(3) Culture and identification of Acanthamoeba
Acanthamoeba keratitis is mostly caused by Acanthamoeba protozoa infection, and there are occasional reports of Negri infection. The culture of Acanthamoeba cornea is conventionally selected page non nutritional agar medium, and its solid medium can be added with 1.5 g agar in 100 ml liquid medium. Before using page non nutritive agar medium, the surface of the agar should be coated with live or inactivated Escherichia coli solution. The specimens should be seeded in the center of the agar, incubated at 28 ℃ in a wet box, and observed every day for at least the 10th day. Acanthamoeba can grow from the inoculation site on the agar surface and move around, producing an irregular track. It can be observed under the microscope after 1 day of culture. After 5 ~ 10 days of inoculation, it can be observed with the naked eye that there are thin corrugated changes on the agar surface, and small round or multilateral thick cysts can be seen under the low power microscope, with strong refraction; The trophozoites are round, oval and irregular, and move around to look for food, so the distal end of the inoculation site is mostly trophozoites. The morphology and structure of Acanthamoeba trophozoites and cysts can be clearly distinguished by cutting a piece of surface agar with a small knife and observing it under the oil microscope of the microscope. Due to the limited anti amoeba drugs, there is no need to carry out routine anti Acanthamoeba drug sensitivity test in clinic. Microdilution method can be used in the development of new drugs.
(4) Nucleic acid detection
At present, there is no nucleic acid amplification test reagent for eye samples approved by the State Drug Administration. Most nucleic acid tests adopt the methods established by the laboratory. For samples with negative routine culture, nucleic acid testing has become the main means to diagnose viral keratitis, uveitis and retinitis. The use of quantitative real-time PCR (qPCR) technology to detect pathogenic microorganisms such as adenovirus, cytomegalovirus, herpes simplex virus, varicella zoster virus, EB virus and Toxoplasma gondii has reached a consensus in the industry [31,32]. This method has high specificity and sensitivity [33]. The infection status and treatment effect can be evaluated by copy number. Its positive predictive value and negative predictive value are better than traditional microbial detection methods [34]. QPCR technology is suitable for acute infection, recovery or transient infection, and false negative results can occur [35]. In addition, 16S rDNA and 18S rDNA sequencing technology can be used to improve the detection rate. In 2017, fluorescence in situ hybridization (FISH) was first used in ophthalmology. Peptide nucleic acid fluorescence in situ hybridization (PNA-FISH) was carried out with Staphylococcus specific molecular peptide nucleic acid (PNA) probe, and the detection of pathogens in intraocular fluid could be realized in 20 minutes [36]. Recently, some studies have expanded the application range of fish and PNA probes, which can quickly detect common bacterial and fungal pathogens of endophthalmitis or keratitis (sensitivity 97%, specificity 100%) [37]. Nucleic acid test specimens shall be placed in closed sterilization containers (such as low-temperature resistant cryopreservation centrifuge tube) immediately after aseptic sampling. The samples to be tested for RNA should be placed in an ice box and sent to the laboratory, and the RNA extraction should be completed within 48 hours; The samples to be tested for DNA can be stored at room temperature, but not more than 72 hours, otherwise they need to be stored at - 20 ℃ or - 80 ℃; In addition, avoid the damage of repeated freezing and thawing of samples to protein samples.
For sterile ocular specimens, if the routine test cannot meet the laboratory diagnosis, metagenomics next generation sequencing (mngs) can be selected. The unbiased nature of this technology makes it an important means of identifying unknown pathogens [38]. Studies have shown that the sensitivity of mngs in the diagnosis of endophthalmitis is 88%. When identifying unknown low abundance microorganisms, mngs is more sensitive than other methods [39]. However, mngs is affected by the amount of samples, sample processing, nucleic acid extraction, library preparation and sequencing depth. The accuracy and specificity of the results need to be further verified in clinic. Non sterile ophthalmic specimens can detect a variety of microorganisms, which is difficult to judge whether they are pathogenic clinically. Despite the limitations of mngs, it is still one of the detection methods of ophthalmic difficult to culture pathogens.
(5) Immunological test
Patients with retinitis or uveitis secondary to systemic infection can undergo serological examination, such as the diagnosis of Treponema pallidum, Toxoplasma gondii and Borrelia burgdorferi. Serum, aqueous humor, vitreous humor and lacrimal sac secretion can be tested for pathogen specific antibodies and total IgG, including herpesvirus, cytomegalovirus, Toxoplasma gondii and syphilis. The methods include enzyme-linked immunosorbent assay (ELISA) Immunofluorescence assay (IFA) and immunoblotting test (IBT), etc. The timing of sample collection is very important for the interpretation of the results. When the recovery period of IgG antibody is 4 times higher than that in the initial stage or significantly higher, and the acute period is significantly higher, it can be judged as positive. It has been reported that after the initial infection with Toxoplasma gondii, IgM and IgA peaked at 7 ~ 14 days after infection, and IgG began to rise at 14 ~ 21 days after infection and lasted for a long time. IgM positive is of certain significance in the diagnosis of early infection. The recovery period of IgG is 4 times higher than that in acute stage, which has retrospective diagnostic significance. The calculation of Goldmann Witmer (GW) coefficient can help judge whether the specific antibody in the eye is produced in situ or false positive due to serum leakage. The coefficient ≥ 4 indicates the production of local antibody, and the coefficient < 2 indicates the loss of local antibody. 2 ~ 3 depends on the clinical situation [27]. In addition, the detection of antibody is related to the immune status of patients. If the serological test result is negative, the infection can be ruled out; However, for those with low immune function, their antibody synthesis ability is limited, and their false negative results need to be carefully explained. The production of antibody is also related to the use of autoantibody, complement and immunosuppressant, so the interpretation of the results must be combined with clinical practice. Several studies have confirmed that the specificity of diagnosis is higher than 99% by combining GW coefficient, Western blot and PCR.
4、 Reporting requirements
The ophthalmic microbiological test report includes sample information, sample processing methods, quality evaluation and microbiological test results (microscopic examination, microbial culture, identification and drug sensitivity test). Based on the expert consensus on the reporting norms of microscopic examination and culture results of bacterial and fungal smears formulated by the branch of laboratory medicine of Chinese Medical Association in 2017 (hereinafter referred to as the 2017 consensus on bacterial and fungal reporting) [40], combined with this consensus, the following requirements for ophthalmic microbiological examination report are put forward.
(1) Specimen information and processing method
The types of common infectious ophthalmopathy specimens include conjunctival sac secretion, corneal focus scraps, palpebral margin or lacrimal passage secretion, anterior chamber aqueous humor, vitreous body, etc. when submitting specimens for examination, the sampling position and sampling method must be accurately marked, such as intraoperative sampling, bedside inoculation, swab transfer, submission time, etc. at the same time, the specimen processing methods, such as centrifugal precipitation, flapping, grinding, etc, And smear staining and culture methods.
(2) Sample quality evaluation results
Follow the above method. Indicate whether the samples are qualified, and the reasons for unqualified samples must be indicated. At present, there is no international quality standard for aqueous humor and vitreous specimens, which can be implemented according to the standard of keratoconjunctival specimens. Since it is difficult to obtain materials, rejection is not recommended even if the number of samples is small. The possible impact on the results can be indicated on the report.
(3) Microscopic examination report
1. Microbiological report: if no microorganism is found in the smear, report: no bacteria, fungi and parasites are found in some staining; If it is positive, report: bacteria or fungi or parasites can be seen in a certain staining, describe the characteristics, quantity and surrounding cytological changes of bacteria, and make a preliminary diagnosis. (1) The description of bacterial characteristics shall include the morphology of bacteria after staining (bacilli, cocci, cocci), staining characteristics (positive, negative or uncertain), and arrangement mode (diplococcus, Staphylococcus, quadruple, etc.). It is suggested to use the semi quantitative reporting method in the 2017 bacterial and fungal reporting consensus, i.e. oil mirror( × 1 000) observation, each visual field ≤ 1 bacterium, recorded as +; 1 ~ 5 bacteria, recorded as + +; 6 ~ 30 bacteria, marked as + + +; > 30 bacteria, recorded as + + + [40]. Give preliminary suspected pathogens. (2) Fungi should describe the morphology, location, size, arrangement and coloring of spores and hyphae respectively, and hyphae should distinguish between true and false hyphae. For hyphae and spores with typical morphological characteristics, suspected microorganisms can be reported; If yeast like spores and pseudohyphae are found, it can be reported that yeast like fungal spores and pseudohyphae are found; Giemsa staining found long filamentous hyphae with septa and 45 ° branches. It can be reported that fungal hyphae with septa and 45 ° branches, suspected Aspergillus, etc. (3) Four stages of Acanthamoeba protozoa can be observed by Giemsa staining or normal saline wet tablets. The morphological characteristics of protozoa should be described in detail in the report, and the number of protozoa should be described with reference to the cell counting method. For mites, how many (number) adults, larvae, eggs / high-power visual field can be reported. Other parasites can be described to give a preliminary diagnosis.
2. Cell classification: in addition to describing microbial characteristics, cell classification shall also be reported, such as keratoconjunctival epithelial cells, pigment epithelial cells, inflammatory exudative cells (neutrophils, eosinophils, mononuclear phagocytes, lymphocytes and plasma cells) and special tissue cells. The relationship between inflammatory cells and pathogens should also be reported. It is suggested to make a graphic report and give a preliminary diagnosis.
3. Cell count: under the visual field of each oil mirror, ≤ 1 cell, recorded as +; Every 1 ~ 5 cells are recorded as + +; 6 ~ 30 cells, marked as + + +; > 30 cells, marked as + + + [40].
4. Microscopic examination result report: it is recommended to carry out the rapid report mode. It is recommended to send the general report within 2 hours, and the complex report should not be sent more than 4 hours. For important pathogens (such as suspected Bacillus cereus or Fusarium, etc.), the critical value is recommended in the report.
(4) Culture and drug sensitivity Report
1. Culture positive report: for the samples with positive culture, it is necessary to further determine whether they are pathogenic microorganisms with reference to the source of the samples. There are normal flora colonization in conjunctiva, eyelid margin and other parts, and colonization bacteria can also lead to infection when immunity is low; The isolation of bacteria from the vitreous and the cornea should be considered. In addition, if you see Gram-positive crude bacillus, you should give a report in time (within 2 hours) to prompt the clinic (the drug should be covered with Bacillus cereus). Finally, the culture results should be comprehensively judged in combination with specimen quality, microscopic examination and clinical diagnosis.
2. Report of negative culture: for the specimen with negative culture, report "how many days after some method of culture, sterile growth" for both the bacterial part and the sterile part. If the cultivation continues after the report is issued, write "how many days the cultivation will continue, this report is a primary report, and how many days after the final result is queried" on the report sheet.
3. Report of suspected contamination: carefully find out the source of contamination for the suspected contaminated isolates. If it is finally determined to be contaminated bacteria in the laboratory, the results should not be reported to the clinic; If the bacterium is contaminated outside the laboratory and it is difficult to determine the source of pollution, it is necessary to communicate with the clinician and truthfully fill in "after how many days of cultivation, it is found that a certain bacterium grows. Because it is impossible to distinguish between pollution and infection, it is recommended to submit it for secondary examination".
4. Report of mixed infection: for mixed infection (i.e. two or more microorganisms are isolated), multiple infections of bacteria, fungi and parasites (such as amoeba) may be involved, especially lacrimal organ infection, where multiple bacteria often coexist. The issuance of mixed infection report should be based on the characteristics of samples and combined with clinical comprehensive judgment.
5. Drug sensitivity report: the eye bacterial drug sensitivity report shall indicate the drug sensitivity judgment standard (i.e. break point) of various antibiotics to be tested. If there is no break point or borrow the break point of other strains and literature, remarks shall be made. At the same time, the version number and publication time of the standard used in the laboratory shall also be given in the report. The ocular bacterial drug sensitivity test includes mic method and disk diffusion method. Therefore, the drug sensitivity report should give the value of mic or the diameter of bacteriostatic circle, and explain the results according to the break point.
(5) Other
Immunological tests are mostly qualitative tests, and the type of methodology should be indicated. Specific nucleic acid amplification includes qPCR and qualitative PCR, which shall be reported according to the instructions. Mngs has diagnostic value for pathogenic microorganisms with negative routine examination. Conditional pathogenic bacteria should be interpreted under the guidance of microbiologists.
5、 Summary
Ophthalmologists should comprehensively evaluate the ocular lesions before applying for examination items, and select staining and culture methods according to clinical diagnosis. If the scraping / smear results are inconsistent with the culture, the laboratory personnel shall communicate with the clinician in time. If the culture of purulent specimens is negative, many factors should be considered, such as material selection, transport mode, transport cycle, use of antibiotics, influence of anesthetics, special pathogens, etc. Here, it is emphasized that ophthalmologists should fully cooperate with microbiological inspectors, collect samples correctly, and advocate bedside vaccination. The laboratory shall establish a technical platform for ophthalmic pathogen detection. In addition to traditional culture, the laboratory shall have detection methods of nucleic acid, antibody and special staining to supplement the inspection contents of viruses, atypical pathogens and parasites. The break point of commonly used antimicrobial susceptibility test is not suitable for local ophthalmic drugs. In most cases, it still needs to rely on doctors' experience or combined with local epidemiological data.
Authors: Wang Zhiqun (Beijing Institute of Ophthalmology, Beijing Tongren Hospital, Capital Medical University), Lu Xinxin (laboratory department, Beijing Tongren Hospital, Capital Medical University), Liang Qingfeng (Beijing Institute of Ophthalmology, Beijing Tongren Hospital, Capital Medical University)
Members of the expert group (sorted by last name Pinyin):
Chen dongke (Laboratory Department of Beijing hospital), Chen Kexin (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to Capital Medical University), Deng Shijing (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to Capital Medical University), Feng Yun (Ophthalmology, the Third Hospital of Peking University), Gu Li (Beijing Institute of respiratory diseases, Beijing Chaoyang Hospital Affiliated to Capital Medical University), Hu Jihong (clinical laboratory center of National Health Commission), Hu Jianzhang (Ophthalmology Department of Union Hospital Affiliated to Fujian Medical University), Huang Yanfei (Laboratory Department of Beijing Tongren Hospital Affiliated to Capital Medical University), Li Zhenjun (Institute for infectious disease prevention and control of China Center for Disease Control and prevention), Lu binghuai (Department of respiratory and critical diseases, China Japan Friendship Hospital), Lu Xinxin (Laboratory Department of Beijing Tongren Hospital Affiliated to Capital Medical University), Liang Lingyi (Department of keratology, Zhongshan Eye Center, Sun Yat sen University), Liang Qingfeng (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to Capital Medical University), Tian Lei (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to Capital Medical University), Tian Xiaobo (Laboratory Department of Beijing Tongren Hospital Affiliated to Capital Medical University), Tao Yong (Ophthalmology Department of Beijing Chaoyang Hospital Affiliated to Capital Medical University), Yang Jiyong (Laboratory Department of the first medical center of PLA General Hospital), Yang peizeng (Ophthalmology Department of the First Affiliated Hospital of Chongqing Medical University), Yuan Xiaoyong (Tianjin Institute of Ophthalmology, Tianjin ophthalmology hospital), Wang Qingtao (Laboratory Department of Beijing Chaoyang Hospital Affiliated to Capital Medical University), Wang Mei (Laboratory Department of Beijing Tongren Hospital Affiliated to Capital Medical University), Wang Zhiqun (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to Capital Medical University), Xu Xizhan (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to capital Medical University), Zhang Yang (Beijing Institute of Ophthalmology, Beijing Tongren Hospital Affiliated to Capital Medical University), Zhao Jianyu (Laboratory Department of Beijing Shunyi District Hospital), Zhou Qianqian (Laboratory Department of Beijing Huilongguan Hospital)
Zhi Xie, Wei Zhenyu (Beijing Institute of Ophthalmology, Beijing Tongren Hospital, Capital Medical University), Wang Leying (Beijing Institute of Ophthalmology, Beijing Tongren Hospital, Capital Medical University), Zhang Zijun (Beijing Institute of Ophthalmology, Beijing Tongren Hospital, Capital Medical University)
References (omitted)